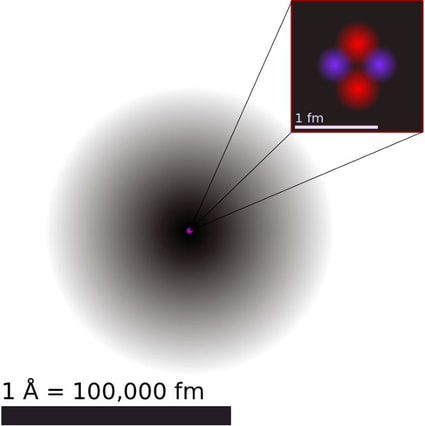
 Genesis 1:2 “And the earth was without form, and void; and darkness was upon the face of the deep. And the Spirit of God moved upon the face of the waters.” Why doesn’t an atom fly apart? It really ought to do so. School children are taught that atoms basically contain three types of particles – protons, neutrons and electrons. The protons and neutrons are in the atomic nucleus. They are roughly the same mass, but neutrons are neutral, whereas protons have a positive electrical charge. Tiny electrons, with a negative charge, whizz around the parent atom at great speed. If you rub a balloon on your hair, you know that you can probably make it stick to a wall. The balloon will pick up a negative electrical charge. Suppose you charge two balloons, each with a string attached. Hold them hanging from your hands, and bring them close. They repel because they have the same charge. The protons in the atomic nucleus have the same positive charge. They should repel. The fact that they do not do so suggests that there is a stronger force present keeping the protons together, even though the electric charge wants to push them apart. This strong force does not have a large sphere of influence. It only seems to operate when objects are 10-15m apart. At such a small distance, the strong force seems to be 137 times stronger than the electrical force. Outside this distance, the electromagnetic force is stronger, so the protons would push each other away. God has balanced these forces exactly. If the strong force were a little weaker, then large atoms would easily fall apart. If a little stronger, then no chemical reactions would occur. creationmoments.com/sermons/forcing-matter-to-stick Comments are closed.
|
Archives
February 2020
|
Ads do not imply endorsement | Policy/Terms of Service | About Us | Contact Us | © Life Work Academy, Inc.
 RSS Feed
RSS Feed

